
Chemistry, 24.09.2019 23:20 bertha4082
The molar absorptivity for aqueous solutions of phenol at 211 nm is 6.17x103l/mol/cm. calculate the linear range of phenol concentrations if the transmittance is to be less than 85% and greater than 7% when the measurements are made in 1cm cell.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:00, melidacampos12
Substance x has a fixed volume, and the attraction between its particles is strong .substance y had widely spread out particles and can be compressed what can most likely be concluded about these substances
Answers: 2

Chemistry, 22.06.2019 01:20, whrjegt4jrnfdvj
1. suppose a reaction mixture, when diluted with water, afforded 300 ml of an aqueous solution of 30 g of the reaction product malononitrile [ch2(cn)2], which is to be isolated by extraction with ether. the solubility of malononitrile in ether at room temperature is 20.0 g/100 ml, and in water is 13.3 g/100 ml. what weight of malononitrile would be recovered by extraction with (a) three 100-ml portions of ether and (b) one 300-ml portion of ether? suggestion: for each extraction, let x equal the weight extracted into the ether layer. in part (a), the concentration in the ether layer is x/100 and in the water layer is (30 x)/300; the ratio of these quantities is equal to k 20/13.3.
Answers: 2

Chemistry, 22.06.2019 03:30, electrofy456
What diagram shows the ionic compound of magnesium oxide
Answers: 2

Chemistry, 22.06.2019 03:30, ilizzy1224
Adrop of acetone (nail polish remover) has a mass of 35 mg and a density of 0.788 g/cm3. what is its volume in cubic centimeters?
Answers: 3
You know the right answer?
The molar absorptivity for aqueous solutions of phenol at 211 nm is 6.17x103l/mol/cm. calculate the...
Questions in other subjects:

Social Studies, 28.11.2019 05:31








Mathematics, 28.11.2019 05:31

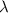 = 211 nm,
= 211 nm, 


 = 1.155
= 1.155
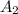 = 0.07058
= 0.07058
 = molar extinction coefficient
= molar extinction coefficient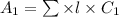
 =
= 


 =
= 




