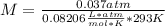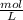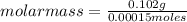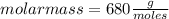Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 17:10, mikeeway33
Some liquids can be distilled, but only at temperatures that are so high that it is impractical, or so high the compound decomposes. explain why distillation such compounds at significantly less than atmospheric pressure (some degree of vacuum) would solve this problem.
Answers: 2

Chemistry, 22.06.2019 20:30, kittybatch345
Is a chemical message sent by another individual.
Answers: 1

Chemistry, 23.06.2019 00:50, alainacorkell6472
What is the enthalpy of combustion (per mole) of c4h10 (g)? –2,657.5 kj/mol –5315.0 kj/mol –509.7 kj/mol –254.8 kj/mol
Answers: 1
You know the right answer?
Asolution containing 0.102 g of an unknown compound dissolved in 100. ml of water has an osmotic pre...
Questions in other subjects:

Chemistry, 21.04.2021 05:40


History, 21.04.2021 05:40




Mathematics, 21.04.2021 05:50



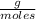
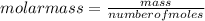
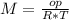
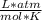 T=20 °C= 293°K (0°C=273 °K)
T=20 °C= 293°K (0°C=273 °K)