
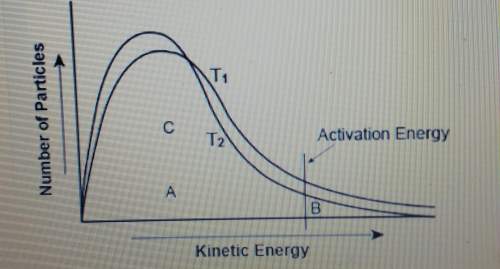
The graph shows the distribution of energy in the particles of 2 gas samples at different temperatures, t1 and t2. a, b, and c represent individual particles.
based on the graph, which of the following statements is likely to be true?
a. particle a is more likely to participate in the reaction than particle b.
b. particle c is more likely to participate in the reaction than particle b.
c. the number of particles able to undergo a chemical reaction is less than the number that is not able to.
d. more gas particles participate in the reaction at t2 than at t1.

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 17:40, aaliyahthomas37
Which statement about hf is true? it is zero for any compound in its standard state. it is positive when the bonds of the product store more energy than those of the reactants. it is negative when a compound forms from elements in their standard states. it is zero for any element that is in the liquid state.
Answers: 1
You know the right answer?
The graph shows the distribution of energy in the particles of 2 gas samples at different temperatur...
Questions in other subjects:

Mathematics, 16.09.2020 02:01

Mathematics, 16.09.2020 02:01

Mathematics, 16.09.2020 02:01

Mathematics, 16.09.2020 02:01

Mathematics, 16.09.2020 02:01

History, 16.09.2020 02:01

Mathematics, 16.09.2020 02:01

Social Studies, 16.09.2020 02:01

English, 16.09.2020 02:01

Mathematics, 16.09.2020 02:01



