
Chemistry, 16.09.2019 21:30 he0gaubong
Which of the following statements about acids and bases are true? acetic acid is a strong acid. a strong acid has a weak conjugate base. a brønsted base is a proton acceptor. bases need not contain –oh directly; instead, they can increase the concentration of –oh by deprotonating water.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:30, thatonestudent2271
If 3.00 g of titanium metal is reacted with 6.00 g of chlorine gas, cl2, to form 7.7 g of titanium (iv) chloride in a combination reaction, what is the percent yield of the product?
Answers: 1

Chemistry, 22.06.2019 14:10, cameronbeaugh
13. a covalent bond between two atoms is likely to be polar if: a. one of the atoms is much more electronegative than the other. b. the two atoms are equally electronegative. c. the two atoms are of the same element. d. the bond is part of a tetrahedrally shaped molecule. e. one atom is an anion.
Answers: 1


Chemistry, 22.06.2019 23:30, johnnysteeler9934
The ammonia molecule in the diagram has the observed bond orientation because
Answers: 1
You know the right answer?
Which of the following statements about acids and bases are true? acetic acid is a strong acid. a st...
Questions in other subjects:


Mathematics, 24.06.2019 13:30


Spanish, 24.06.2019 13:30

Social Studies, 24.06.2019 13:30

History, 24.06.2019 13:30

Chemistry, 24.06.2019 13:30


History, 24.06.2019 13:30

Business, 24.06.2019 13:30

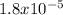 , pKa will be
, pKa will be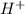 and
and 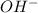 directly, they can increase their concentration by deprotonating water.
directly, they can increase their concentration by deprotonating water. 

