
Chemistry, 14.09.2019 11:10 amiechap12
1. make a 100 ml volume of 100 mm acetic acid (hac) hac (glacial, 100%, 17.4 m, 1.05 g/ml, mw 60.05) ddh,0 ml add to 100 ml measure 250 ml beaker. add ml of ddh20 using a 100-ml graduated cylinder and transfer this volume to a ul of 100 mm hac to the water. mix with a stir bar.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:30, portedon8644
13. calculate the initial concentration (before precipitation) of carbonate ions after the addition of each 0.05 ml of solution b to the 1.00 l beaker of solution a. divide the work among group members and write the answers in the table in model 3. assume the volume change as solution b is added is negligible. 14. notice the initial concentrations of zn2+ - and cu2+ in the table in model 3. a. explain how these were obtained from the data in model 2. b. as solution b is added and precipitates form, do these initial concentrations change? 15. use the data in model 2 to indicate the presence of precipitate (either znco3 or cuco3) after each 0.05 ml addition of solution b in model 3. 16. use the initial concentrations of carbonate ions and zinc ions to calculate the reaction quotient, qsp for the zinc carbonate scenarios in model 3. divide the work among group members and write the answers in the table in model 3. 17. use the initial concentrations of carbonate ion and copper(ii) ions to calculate the qsp for the copper(ii) carbonate scenarios in model 3. divide the work among group members and write the answers in the table in model 3.
Answers: 3



Chemistry, 22.06.2019 14:10, roserose3098
16. in a reaction that has reached equilibrium, a. the forward and reverse reactions are occurring at the same rate. b. the reactants and products are in equal concentrations. c. the forward reaction has gone further than the reverse reaction. d. there are equal numbers of atoms on both sides of the equation. e. a, b, and d are correct.
Answers: 2
You know the right answer?
1. make a 100 ml volume of 100 mm acetic acid (hac) hac (glacial, 100%, 17.4 m, 1.05 g/ml, mw 60.05)...
Questions in other subjects:

History, 10.03.2020 23:05






History, 10.03.2020 23:05


Mathematics, 10.03.2020 23:05

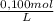 = 0,0100 moles of acetic acid
= 0,0100 moles of acetic acid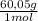 ×
×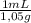 = 0,57 mL of glacial acetic acid
= 0,57 mL of glacial acetic acid


