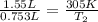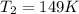If the volame ofa gas coetainer at 32°c changes froem 1.55 l to 753 ml, what will the final temperature be? assume the pressure doesn't change and the amount of gas in the coatainer doesn't change. remember that there are 1,000 ml in 1 l. o a. 149°c ob. 353 c oc. 273c o d.-125 c oe none of the above.

Answers: 3


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 18:50, cj31150631
Question 3(multiple choice worth 4 points) (04.04 lc) what does it mean when an element is reduced? it empties a valance shell, reducing its atomic radius. it gains electrons, reducing its overall charge. it increases electronegativity, reducing its ability to bond. it loses electrons, reducing its electron number.
Answers: 1

Chemistry, 22.06.2019 20:00, 20calzoy
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. in the first step, calcium carbide and water react to form acetylene and calcium hydroxide: cac2 (s) + 2h2o (g) → c2h2 (g) + caoh2 (s) =δh−414.kj in the second step, acetylene, carbon dioxide and water react to form acrylic acid: 6c2h2 (g) + 3co2 (g) + 4h2o (g) → 5ch2chco2h (g) =δh132.kj calculate the net change in enthalpy for the formation of one mole of acrylic acid from calcium carbide, water and carbon dioxide from these reactions. round your answer to the nearest kj .
Answers: 3
You know the right answer?
If the volame ofa gas coetainer at 32°c changes froem 1.55 l to 753 ml, what will the final temperat...
Questions in other subjects:


Physics, 03.07.2020 17:01

Mathematics, 03.07.2020 17:01




English, 03.07.2020 17:01



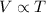
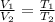
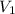 = initial volume of gas = 1.55 L
= initial volume of gas = 1.55 L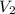 = final volume of gas = 753 ml = 0.753 L
= final volume of gas = 753 ml = 0.753 L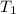 = initial temperature of gas =
= initial temperature of gas = 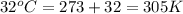
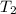 = final temperature of gas = ?
= final temperature of gas = ?