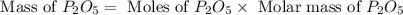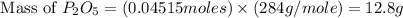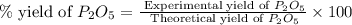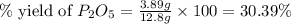Chemistry, 14.09.2019 07:30 holycow7868
Determine the percent yield of the following reaction when 2.80 g of p reacts with excess oxygen. the actual yield of this reaction is determined to by 3.89 g of p2o5.
4 p + 5 o2 > 2 p2o5

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:00, mbrisen7420
Compare and contrast physical changes with chemical changes.
Answers: 3

Chemistry, 22.06.2019 09:30, raizagisselle1694
Mike and mitchell decide to have a foot race. they mark off a stretch of 100 yards, and recruit cindy to work the stopwatch. after running the race and looking at the results, cindy declared that mitchell was the fastest. so how did the boys times compare?
Answers: 3

Chemistry, 22.06.2019 12:00, BreBreDoeCCx
Ageochemist examines a piece of metal that he found in the soil. he performs tests to identify the metal from its density, electrical conductivity, and melting point. which statement best describes his investigation? a. he is determining physical properties that are sufficient to identify the metal. b. he is determining chemical properties that are sufficient to identify the metal. c. he is determining physical properties that are insufficient to identify the metal. d. he is determining chemical properties that are insufficient to identify the metal.
Answers: 3
You know the right answer?
Determine the percent yield of the following reaction when 2.80 g of p reacts with excess oxygen. th...
Questions in other subjects:



Chemistry, 04.09.2020 01:01






Biology, 04.09.2020 01:01

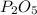 is, 30.39 %
is, 30.39 %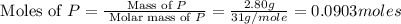
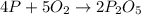
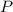 react to give 2 mole of
react to give 2 mole of 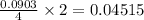 moles of
moles of