
Chemistry, 14.09.2019 07:30 qdogisbeast6132
For the following reaction, calculate how many grams of each product are formed when 4.05 g of water is used.
2 h20 > 2 h2 + o2

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:50, giiffnlojd
Using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 pb+2(aq) + 2cl -(aq). if the value of ksp was determined to be only 1.2 x 10-2: too much solid has dissolved. additional precipitate is forming. the solution is unsaturated. the ions are now combining to reduce their concentrations.
Answers: 3



You know the right answer?
For the following reaction, calculate how many grams of each product are formed when 4.05 g of water...
Questions in other subjects:


English, 26.09.2019 03:10



English, 26.09.2019 03:20


Mathematics, 26.09.2019 03:21


English, 26.09.2019 03:21

English, 26.09.2019 03:21

 .....(1)
.....(1)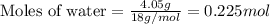

 of hydrogen gas.
of hydrogen gas.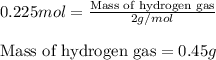
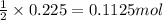 of nitrogen gas.
of nitrogen gas.


