
Chemistry, 14.09.2019 06:30 Averyruss9245
What is the ph of a 0.18 m ch3nh3+cl– aqueous solution? the pkb ofch3nh2 is 3.44

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:10, Kianna000
56.16 gregor mendel was the first scientist to use statistics to analyze scientific data. before mendel's experiments, scientists believed that organisms acquired traits from their environment and passed them on to their offspring. after mendel's discoveries were accepted, scientists realized that traits passed to offspring were the result of genes being passed from parents to offspring. this is an example of pls
Answers: 1

Chemistry, 22.06.2019 09:20, pandaman632
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1

Chemistry, 22.06.2019 10:30, Clivensp5
Skills of homo sapiens were found an excavation. the skulls were preserved because the bodies were frozen. so, these fossils are (blank) fossils. the image shows the evolution of skulls beginning 2 to 3 million years ago. based on the image, modern human skulls(blank) ape skulls.
Answers: 1

Chemistry, 22.06.2019 22:30, teagan56
Gusing the milligrams of ascorbic acid you entered above, the ratio of total sample volume to aliquot volume, and the total milligrams of the vitamin c tablet that you dissolved, calculate the mass of ascorbic acid in the vitamin c tablet for each trial. do this by scaling up to find the amount (mg) of ascorbic acid in your 250 ml flask. enter your calculated mass of ascorbic acid in the vitamin c tablet, for each trial. be sure to enter your calculated mass in the corresponding order that you entered your milligrams of ascorbic acid. the milligrams of ascorbic acid you entered for entry #1 previously should correspond to the mass of ascorbic acid that you enter for entry #1 here.
Answers: 1
You know the right answer?
What is the ph of a 0.18 m ch3nh3+cl– aqueous solution? the pkb ofch3nh2 is 3.44...
Questions in other subjects:

Mathematics, 17.10.2019 07:30

Mathematics, 17.10.2019 07:30

Mathematics, 17.10.2019 07:30

Mathematics, 17.10.2019 07:30


Social Studies, 17.10.2019 07:30

English, 17.10.2019 07:30






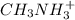 ⇄ H⁺ +
⇄ H⁺ + 
![K_{a}=\frac {\left [ H^{+} \right ]\left [CH_3NH_2 \right ]}{[CH_3NH_3^+]}](/tpl/images/0230/9864/9c094.png)





