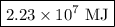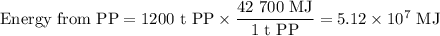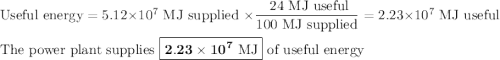The heat of combustion of pp is 42.7 mj kg1. if pp containers are urned in a waste-fired power station that generates electricity with an verall efficiency of 24%, calculate how much useful electrical energy generated when 1,200 tonnes of pp is burned. give your answer in nits of mj to 3 significant figures and explain your working

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 23.06.2019 09:20, taylorannsalazar
La reaccion entre monoxido de nitrogeno (no) y oxigeno para formardioxido de nitrogeno (no2) es un paso determinante para la formacion del smog, la reaccion es la siguiente: 2no + o2 = 2no2 cual sera el numero de moles de no2 que se formaran por la reaccion completa de 8 moles de oxigeno con suficiente monoxido?
Answers: 1

You know the right answer?
The heat of combustion of pp is 42.7 mj kg1. if pp containers are urned in a waste-fired power stati...
Questions in other subjects:

Mathematics, 24.09.2021 14:20


English, 24.09.2021 14:20


Mathematics, 24.09.2021 14:20


Chemistry, 24.09.2021 14:20

Mathematics, 24.09.2021 14:20


Chemistry, 24.09.2021 14:20