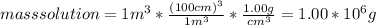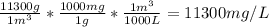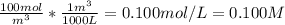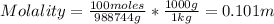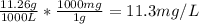Chemistry, 13.09.2019 22:30 maddoxlachowski
The concentration of chlorobenzene (c& hscl) in water is 100 mol/m3. density is 1.00 g/cm3 the solution (a) what is the weight fraction of chlorobenzene? (b) what is the chlorobenzene concentration in ppm? (c) what is the mole fraction of chlorobenzene? (d) what is the molarity of chlorobenzene? (e) what is the molality of chlorobenzene? the concentration of chlorobenzene (c& hscl) in air is 0.100 mol/m3 at 25 °c and 1 atm. the molecular weight of air may be taken to be 28.84 gmol. (a) what is the weight fraction of chlorobenzene? (c) what is the mole fraction of chlorobenzene? (b) what is the chlorobenzene concentration in ppm?

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 13:00, keananashville
Metallic bonds are good conductors of electricity true or false
Answers: 2

Chemistry, 21.06.2019 18:30, jadepotts3965
Calculate the change in entropy if br2(l) is converted into gaseous br atoms. s° for br2(l) = 152.2 j/(mol•k) s° for br2(g) = 245.5 j/(mol•k) s° for br(g) = 175.0 j/(mol•k)
Answers: 2

Chemistry, 22.06.2019 05:50, mrylenastewart
What are transitions between a liquid and a solid called? identify which way they are transitioning
Answers: 2

Chemistry, 22.06.2019 06:00, Chente379
An atom of lithium (li) and an atom of chlorine (cl) engage in a chemical reaction. which correctly describes the structure of the resulting chemical compound? hint: consider the class of each element. the chemical compound will have a network structure. the chemical compound will have triple bonds. the chemical compound will have a ball-and-stick structure. the chemical compound will have double bonds.
Answers: 2
You know the right answer?
The concentration of chlorobenzene (c& hscl) in water is 100 mol/m3. density is 1.00 g/cm3 the s...
Questions in other subjects:

History, 29.11.2019 06:31

Mathematics, 29.11.2019 06:31

Social Studies, 29.11.2019 06:31





Chemistry, 29.11.2019 06:31

Mathematics, 29.11.2019 06:31

Computers and Technology, 29.11.2019 06:31