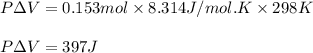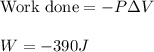Chemistry, 13.09.2019 20:30 willcohen42
Apellet of zn of mass 10.0g is dropped into a flaskcontaining
dilute h2so4 at a pressure of p=1.00 bar and
temperature of 298k. what is the reaction thatoccures? calculate w
for the process.

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 23.06.2019 01:00, MrTeriffic
Imagine if during the cathode ray experiment, the size of the particles of the ray was the same as the size of the atom forming the cathode. which other model or scientific observation would have also been supported? 1. this would support dalton's postulates that proposed the atoms are indivisible because no small particles are involved. 2. this would support bohr's prediction about electrons moving in orbits having specific energy. 3. this would support bohr's prediction about electrons being randomly scattered around the nucleus in the atom. 4. this would support dalton's postulates that proposed that atoms combine in fixed whole number ratios to form compounds.
Answers: 1

Chemistry, 23.06.2019 04:20, vliu732
The reaction below shows a system in equilibrium. how would a decrease in temperature affect this reaction? a. the rate of formation of the gases would increase. b. the equilibrium of the reaction would shift to the left. c. the equilibrium would shift to cause the gases to sublime into solids. d. the chemicals on the left would quickly form the chemical on the right.
Answers: 1

Chemistry, 23.06.2019 08:00, george27212
What is the temperature in kelvin of a gas if it is allowed to expand from 1.50 l to 4.50 l? the initial temperature is 10.0°c and pressure is constant throughout the change. which equation should you use? t2= v2/v1 t1 what is the final temperature? ⇒ 849 k these are the answers.
Answers: 1
You know the right answer?
Apellet of zn of mass 10.0g is dropped into a flaskcontaining
dilute h2so4 at a pressure of p=...
dilute h2so4 at a pressure of p=...
Questions in other subjects:

Computers and Technology, 25.03.2021 06:20

Mathematics, 25.03.2021 06:20


Social Studies, 25.03.2021 06:20

Geography, 25.03.2021 06:20


Mathematics, 25.03.2021 06:20

Mathematics, 25.03.2021 06:20


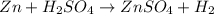
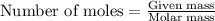
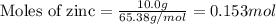
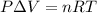
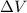 = Change in volume of the gas
= Change in volume of the gas