
Which of the following choices has the compounds correctly arranged in order of increasing solubility in water? (least soluble to most soluble) which of the following choices has the compounds correctly arranged in order of increasing solubility in water? (least soluble to most soluble) lif < nano3 < chcl3 ch3oh < ch4 < lif ch4 < nano3 < chcl3 ccl4 < chcl3 < nano3 ch3oh < cl4 < chcl3

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:00, gilbert325
Write the chemical symbols for three different atoms or atomic cations with 27 electrons. asap!
Answers: 2

Chemistry, 22.06.2019 03:40, jude3412
In an effort to address concerns about global warming, a power plant in portland, oregon is designed to take all of its exhaust gases from its boilers and recycle the co2 using the solvay process to make sodium hydrogen carbonate. the reaction is shown below. nh3(g) + h2o(l) + co2(g) + nacl(aq) → nahco3(aq) + nh4cl(aq) how many liters each of nh3 and co2 (both at stp) would be consumed to produce 3.00 kg of sodium bicarbonate? the volume of both nh3 and co2 would be
Answers: 1

Chemistry, 22.06.2019 10:00, berniceallonce22
What is the atomic mass of an atom that has 6 protons, 6 neutrons, and 6 electrons? a) 6 b) 8 c) + 1 d) 12 e) 18
Answers: 1
You know the right answer?
Which of the following choices has the compounds correctly arranged in order of increasing solubilit...
Questions in other subjects:

Spanish, 17.02.2021 19:40


English, 17.02.2021 19:40


Computers and Technology, 17.02.2021 19:40




Business, 17.02.2021 19:40

Social Studies, 17.02.2021 19:40

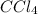 is a non-polar compound. Hence, it is insoluble in water.
is a non-polar compound. Hence, it is insoluble in water. 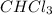 is a polar compound due to difference in electronegativity of chlorine and carbon atom there will be development of partial charges. Hence, there will be dipole-dipole forces existing between them.
is a polar compound due to difference in electronegativity of chlorine and carbon atom there will be development of partial charges. Hence, there will be dipole-dipole forces existing between them. 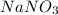 is an ionic compound and it will readily dissociate into ions when dissolved in water. Also, there will be ion-dipole interactions between sodium and nitrate ions.
is an ionic compound and it will readily dissociate into ions when dissolved in water. Also, there will be ion-dipole interactions between sodium and nitrate ions. 

