Chemistry, 11.09.2019 00:10 Trucofer8159
Jim, jill, robert, and kim each run paper chromatography on an unknown aqueous mixture. jim gets a red band and a blue band, jill gets a yellow band and a blue band, robert gets a green band, and kim gets a red band and a yellow band.
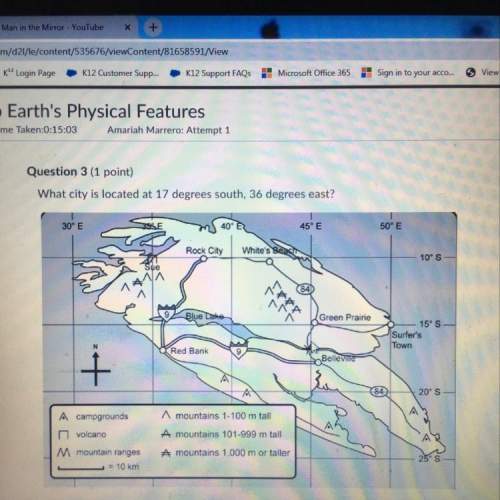
which student did not have more than one substance dissolved in the water?
robert
kim
jill
jim

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 18:30, chinadoll24
Asample of hydrated tin (ii) chloride (sncl2) has a mass of 4.90 g. when it is dehydrated, it has a mass of 4.10 g. which is the correct chemical formula for the hydrate? sncl2•2h2o sncl2•4h2o sncl2•6h2o
Answers: 2

Chemistry, 23.06.2019 04:31, laurenbreellamerritt
How big are the bighest ocean waves at mavericks
Answers: 1

Chemistry, 23.06.2019 09:30, Adolfosbaby
If the solubility of a gas in water is 1.22g/2.75 atm, what is it’s solubility (in g/l) at 1.0 atm
Answers: 1
You know the right answer?
Jim, jill, robert, and kim each run paper chromatography on an unknown aqueous mixture. jim gets a r...
Questions in other subjects:

Mathematics, 05.07.2019 00:00

History, 05.07.2019 00:00

Mathematics, 05.07.2019 00:00


Mathematics, 05.07.2019 00:00

Social Studies, 05.07.2019 00:00

Computers and Technology, 05.07.2019 00:00

Arts, 05.07.2019 00:00

Spanish, 05.07.2019 00:00