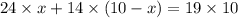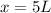Chemistry, 09.09.2019 17:20 kerryjenkins300
A24% acid solution is mixed with a 14% acid solution to form a 19% acid solution. how many liters of the 24% acid solution are used to make 10 liters of the 19% acid solution?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 12:30, kat2177
The electron configurations of two different atoms are shown below. each yellow electron has a charge of 1−, and the net charge of each nucleus is shown. these atoms will combine with bond. a. an ionic b. a positive c. a negative d. a covalent plzzz mee with !
Answers: 1



Chemistry, 22.06.2019 05:30, MansellS5529
A3.37-mg sample of protein was chemically digested to convert its nitrogen into ammonia and then diluted to 100.0 ml. then 10.0 ml of this solution was placed in a 50-ml volumetric flask and treated with 5 ml of phenol solution plus 2 ml of sodium hypochlorite solution. the sample was diluted to 50.0 ml, and the absorbance at 625 nm was measured in a 1.00-cm cuvette and found to be 0.486. for reference, a standard solution was prepared from 10.0 mg of nh4cl (molar mass = 53.49 grams/mole) dissolved in 1.00 l of water. then 10.0 ml of this standard was placed in a 50-ml volumetric flask, treated in the same manner as the unknown, and the absorbance found to be 0.323. finally, a reagent blank was prepared using distilled water in place of unknown, it was treated in the same manner as the unknown, and the absorbance found to be 0.076. calculate the weight percent of nitrogen in the protein.
Answers: 1
You know the right answer?
A24% acid solution is mixed with a 14% acid solution to form a 19% acid solution. how many liters of...
Questions in other subjects:

Mathematics, 18.11.2020 23:00


Mathematics, 18.11.2020 23:00

Physics, 18.11.2020 23:00

Mathematics, 18.11.2020 23:00

Mathematics, 18.11.2020 23:00


Mathematics, 18.11.2020 23:00


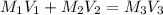
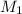 = concentration of first solution = 24%
= concentration of first solution = 24% 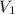 = volume of first solution = x L
= volume of first solution = x L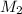 = concentration of second solution = 14%
= concentration of second solution = 14%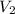 = volume of second solution = (10-x) L
= volume of second solution = (10-x) L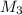 = concentration of final solution = 19%
= concentration of final solution = 19%