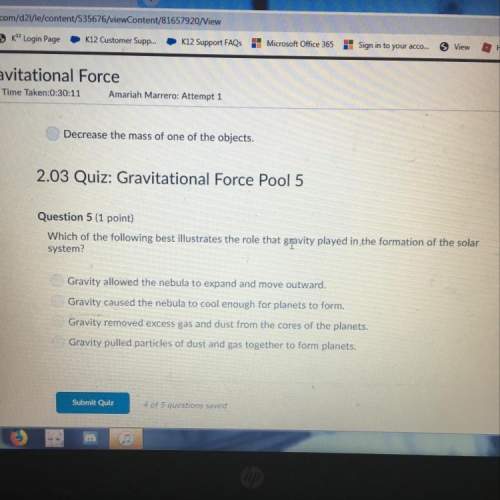
Calculate δgo for the reaction of ammonia with oxygen:
4nh3 (g) + 3 o2 (g) → 2n2 (g) + 6 h2o...

Chemistry, 06.09.2019 20:30 zacksoccer8279
Calculate δgo for the reaction of ammonia with oxygen:
4nh3 (g) + 3 o2 (g) → 2n2 (g) + 6 h2o (g)
using the following data:
substance nh3 (g) o2 (g) n2 (g) h2o (g)
δgo (kj/mol) - 16 0 0 -228.6
a) 212.6 kj
b) 1307.6 kj
c) - 212.6 kj
d) -1435.6 kj
e) -1307.6kj

Answers: 1


Other questions on the subject: Chemistry



You know the right answer?
Questions in other subjects:


Social Studies, 09.12.2021 02:40





Biology, 09.12.2021 02:40



Social Studies, 09.12.2021 02:40