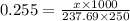Chemistry, 06.09.2019 04:10 valeriekbueno
How many grams of nickel (ii) chloride hexahydrate are required to prepare 250. ml of aqueous solution whose concentration is 0.255 m? the molar mass of nickel (ii) chloride hexahydrate is 237.69 g mol−1.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:00, deidaralove90
Look at the bean data from days 4–6. use these data to explain how natural selection changed the number of dark red walking beans over time. writing part
Answers: 3

Chemistry, 22.06.2019 09:00, mercymain1014
An excess of lithium oxide undergoes a synthesis reaction with water to produce lithium hydroxide li2o+h2o→2lioh if 1.05 g of water reacted, what is the theoretical yield of lithium hydroxide? a) 5.83 x 10–2 g lioh b) 1.17 x 10–1 g lioh c) 2.79 x 100 g lioh d) 1.40 x 100 g lioh
Answers: 1

Chemistry, 22.06.2019 12:30, meghan2529
The melting point of sulfur is 115 °c and its boiling point is 445 °c. what state would sulfur be in at 200 °c?
Answers: 1
You know the right answer?
How many grams of nickel (ii) chloride hexahydrate are required to prepare 250. ml of aqueous soluti...
Questions in other subjects:

Mathematics, 09.04.2020 01:41

Mathematics, 09.04.2020 01:41






Law, 09.04.2020 01:41

Mathematics, 09.04.2020 01:41

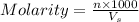
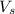 = volume of solution in ml
= volume of solution in ml