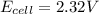The standard emf for the cell using the overall cell reaction below is +2.20 v:
2al(s) + 3i2...

Chemistry, 06.09.2019 02:30 kennycooper3096
The standard emf for the cell using the overall cell reaction below is +2.20 v:
2al(s) + 3i2(s) → 2al3+(aq) + 6i-(aq)
the emf generated by the cell when [al3+] = 4.5 × 10-3 m and [i-] = 0.15 m is v.
(a) 2.23
(b) 2.20
(c) 2.39
(d) 2.32
(e) 2.10

Answers: 2


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 06:30, AleciaCassidy
Identify the missing numbers below to show the result of multiplying the numbers (1.6 × 10-19)(5.0 × 106) = c × 10d
Answers: 1

Chemistry, 22.06.2019 10:30, angemango3423
What is the empirical formula of c6h18o3? ch3o c2h5o c2h6o c2h5o5
Answers: 1
You know the right answer?
Questions in other subjects:

History, 26.09.2019 04:00

Computers and Technology, 26.09.2019 04:00




Chemistry, 26.09.2019 04:00

Social Studies, 26.09.2019 04:00

History, 26.09.2019 04:00



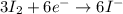
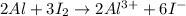
 -
-![E_{cell}=E_{cell}^{0}-\frac{0.059}{n}log{[Al^{3+}]^{2}[I^{-}]^{6}}](/tpl/images/0224/1322/6e080.png)
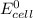 is standard cell emf ,
is standard cell emf , 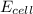 is cell emf ,
is cell emf , ![[Al^{3+}]](/tpl/images/0224/1322/0029f.png) is concentration of
is concentration of 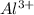 and
and ![[Cl^{-}]](/tpl/images/0224/1322/fd42d.png) is concentration of
is concentration of 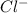
![E_{cell}=2.20-\frac{0.059}{6}log[(4.5\times 10^{-3})^{2}\times (0.15)^{6}]V](/tpl/images/0224/1322/f3bfb.png)