Answers: 3


Other questions on the subject: Chemistry


Chemistry, 23.06.2019 14:50, aleilyg2005
Use the bond energies provided to estimate δh°rxn for the reaction below. ch3oh(l) + 2 o2(g) → co2(g) + 2 h2o(g) δh°rxn = ? bond bond energy (kj/mol) c-h 414 c-o 360 c=o 799 o=o 498 o-h 464 use the bond energies provided to estimate δh°rxn for the reaction below. ch3oh(l) + 2 o2(g) → co2(g) + 2 h2o(g) δh°rxn = ? bond bond energy (kj/mol) c-h 414 c-o 360 c=o 799 o=o 498 o-h 464 +473 kj +206 kj -392 kj -91 kj -486 kj
Answers: 1

Chemistry, 23.06.2019 16:30, mbonham481
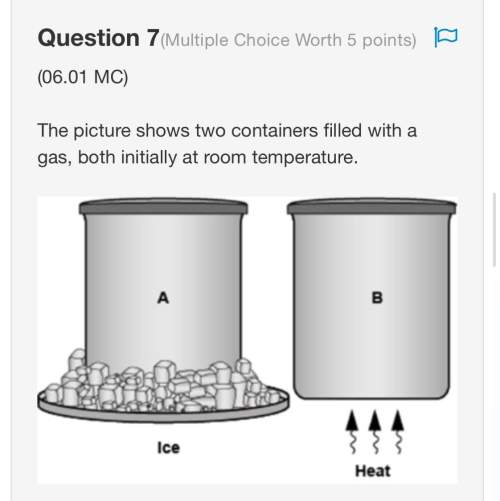
Amodel of an atom is shown below. which element is represented by this model of an atom? boron, carbon, neon, or sodium?
Answers: 1

Chemistry, 23.06.2019 18:00, keshewar4427
Amolecule is a(n) - a. element that physically combines with another element. b. particle composed of two or more atoms bonded together covalently. c. element that isn't bonded to another element.
Answers: 1
You know the right answer?
Battery acid has a density of 1.285 g/ml and contains 38.0% sulfuric acid by mass. the car battery c...
Questions in other subjects:



English, 05.09.2020 01:01




Social Studies, 05.09.2020 01:01


Business, 05.09.2020 01:01

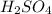 is dissolved in 100 g of solution.
is dissolved in 100 g of solution.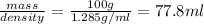
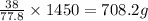 of
of