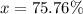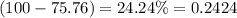Chemistry, 05.09.2019 16:30 lavander9303
Only two stable isotopes of chlorine exist. 35cl has a mass of 34.96885 amu and 37cl has a mass of 36.9659 amu. given that the atomic mass of chlorine is 35.453, calculate the isotopic abundance of 37cl. (enter your answer as a decimal number between zero and one.)

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:30, nxusasmangaliso8780
Which sentence best describes the formation of igneous rock? a- lava on the surface dries up and makes arock b_melted rocks cools and forms crystals c_rocks under tremendous heat and pressure d_magma is melted rock underground
Answers: 1


Chemistry, 22.06.2019 13:00, wbrandi118
Using the thermodynamic information in the aleks data tab, calculate the standard reaction free energy of the following chemical reaction: →+p4o10s6h2ol4h3po4s round your answer to zero decimal places.
Answers: 3

Chemistry, 22.06.2019 19:30, xxaurorabluexx
Chlorine and water react to form hydrogen chloride and oxygen, like this: 2cl2 (g) + 2h2o (g) → 4hcl (g) + o2 (g) also, a chemist finds that at a certain temperature the equilibrium mixture of chlorine, water, hydrogen chloride, and oxygen has the following composition: compound concentration at equilibrium cl2 0.55m h2o 0.57m hcl 0.53m o2 0.34m calculate the value of the equilibrium constant kc for this reaction. round your answer to 2 significant digits.
Answers: 2
You know the right answer?
Only two stable isotopes of chlorine exist. 35cl has a mass of 34.96885 amu and 37cl has a mass of 3...
Questions in other subjects:




English, 21.12.2020 20:20



History, 21.12.2020 20:20

Mathematics, 21.12.2020 20:20


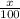
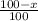
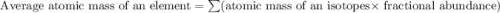
![35.453=\sum[(34.96885 \times \frac{x}{100})+(36.9659 \times \frac{100-x}{100}]]](/tpl/images/0223/4977/3a756.png)