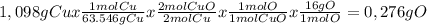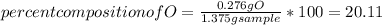Chemistry, 05.09.2019 16:10 alyssatamayo641
When 1.375 g of copper (ii) oxide is reduced on heating in a current of hydrogen, the weight of copper remaining after the reaction is complete is 1.098 g. what is the percent composition of oxygen?

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:30, nuclearfire278
Why is soap used to remove grease? a. its nonpolar end dissolves the grease. b. it makes the water bond with the grease. c. it chemically bonds with the grease. d. its polar end dissolves the grease. correct answer for apex - a, its nonpolar end dissolves the grease.
Answers: 1

Chemistry, 22.06.2019 08:00, ggdvj9gggsc
Asap! will give brainiest when a heat wave strikes a region causing more people to run air-conditioning units, electrical demand increases. what needs to be done to meet this increased demand? raising the control rodslowering the control rodsremoving the control rods
Answers: 1

Chemistry, 22.06.2019 10:10, andersonemma2222
How do you identify the anode on a power source such as a battery? how do you identify the cathode? how are terms anion and cation?
Answers: 1

Chemistry, 22.06.2019 10:30, Brookwiggington8814
What woukd most likely be the transmittance at a 0.70 m solution of solute a? a) 7.6%b) 1.1%c)4.0%d)4.6%
Answers: 1
You know the right answer?
When 1.375 g of copper (ii) oxide is reduced on heating in a current of hydrogen, the weight of copp...
Questions in other subjects:

History, 05.05.2020 07:47

Mathematics, 05.05.2020 07:47




History, 05.05.2020 07:47


Mathematics, 05.05.2020 07:47

Mathematics, 05.05.2020 07:47