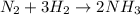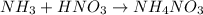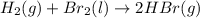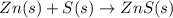Chemistry, 02.09.2019 21:30 Spencerg325
Write balanced chemical equation for the reactions used to prepare each of the following compounds from the given starting material(s). in some cases, additional reactants may be required.
(a) solid ammonium nitrate from gaseous molecule of nitrogen viaa two step process (first reduce the nitrogen to ammonia than neutrlize the ammonium in an appropriate acid)
(b)gaseous hydrogen bromide liquid molecular bromin via one step redox reaction
(c)gaseous h2s from solid zn and s via a two step process(first a redox reaction between the starting material then reaction of the product with a strong acid)

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 23.06.2019 00:50, maddysmall32
Which of the following warnings would an agricultural chemist tell a farmer who wants to recycle his or her own ammonia? recycling ammonia is a difficult process that sometimes takes weeks. recycling ammonia requires a degree in biochemistry or a related field. recycling ammonia can be harmful because it is highly flammable and toxic. recycling ammonia costs too much money considering the price of the necessary chemicals.
Answers: 1

Chemistry, 23.06.2019 07:50, alexusnicole817
Asolution is produced in which water is the solvent and there are four solutes. which of the solutes can dissolve better if the solution is heated?
Answers: 1

Chemistry, 23.06.2019 08:50, leah5981
Reacting masses1 calcium carbonate breaks down on heating to produce calcium oxide and carbondioxide gas. caco3 + cao + co2a student heats 15 g of calcium carbonate strongly in a crucible. relative atomic masses (a): ca = 40, c = 12, o = 16.calculate the mass of calcium oxide produced by this reaction.(5 marks)
Answers: 3
You know the right answer?
Write balanced chemical equation for the reactions used to prepare each of the following compounds f...
Questions in other subjects:

Chemistry, 09.04.2020 02:51



Social Studies, 09.04.2020 02:51



Mathematics, 09.04.2020 02:51


Chemistry, 09.04.2020 02:51

English, 09.04.2020 02:51