Chemistry, 02.09.2019 20:10 allsherm23
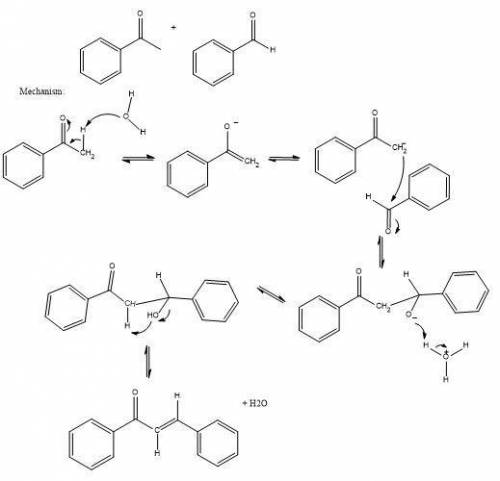
Q3. 3 explain with a detailed mechanism, why a b-hydroxyketone is not isolated as the final product in the base catalysed condensation of acetophenone and benzaldehyde. (5)

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 15:30, lizzyhearts
Count the number of each type of atom in the equation below, and then balance the equation. write in the numbers of atoms and coefficients. add a 1 if there should be no coefficient. cs2(l) + o2(g) → co2(g) + so2(g) c [ ] s [ ] o > c [ ] s [ ] o [ ] cs2(l) + [ ] o2(g) > [ ] co2(g) + [ ] so2(g)
Answers: 3

Chemistry, 22.06.2019 18:00, jeepjose58
What amount of heat is exchanged when 106.2 grams of substance y goes from a liquid at 35 degrees celsius to a solid at the same temperature? melting point of substance y = 35 degrees c; δhvaporization = 3.67 j/mol; δhfusion = 3.30 j/mol. mwsubstance y = 28.22 g/mol. −12.4 j −3.51 x 102 j 1.24 x 101 j 351 j
Answers: 1

You know the right answer?
Q3. 3 explain with a detailed mechanism, why a b-hydroxyketone is not isolated as the final product...
Questions in other subjects:







Mathematics, 19.09.2020 01:01

Mathematics, 19.09.2020 01:01

Chemistry, 19.09.2020 01:01

Health, 19.09.2020 01:01