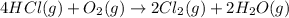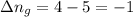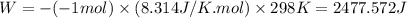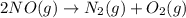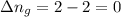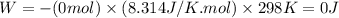Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:30, luhmimi17
Melting and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 1

Chemistry, 22.06.2019 09:40, keiracoles
How many grams of aluminum will there be in 98g of al2o3?
Answers: 1

Chemistry, 22.06.2019 23:10, carmenguabaoql9kv
Afusion reaction takes place between carbon and another element. neutrons are released, and a different element is formed. the different element is a) lighter than helium. b)heavier than helium. c)the same weight as helium. d)dependent on the element that reacted with carbon.
Answers: 3

Chemistry, 23.06.2019 00:50, alainacorkell6472
What is the enthalpy of combustion (per mole) of c4h10 (g)? –2,657.5 kj/mol –5315.0 kj/mol –509.7 kj/mol –254.8 kj/mol
Answers: 1
You know the right answer?
11. assuming that the gases are ideal, calculate the amount of work done (joules) in each of the fol...
Questions in other subjects:

Mathematics, 17.09.2019 05:10


Mathematics, 17.09.2019 05:10

Mathematics, 17.09.2019 05:10

Mathematics, 17.09.2019 05:10

English, 17.09.2019 05:10


Geography, 17.09.2019 05:10

Mathematics, 17.09.2019 05:10

Mathematics, 17.09.2019 05:10

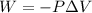
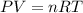
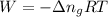 ......(1)
......(1)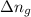 = difference in number of moles of products and reactants =
= difference in number of moles of products and reactants = 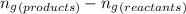
![25^oC=[273+25]K=298K](/tpl/images/0221/2455/0e82f.png)