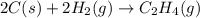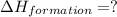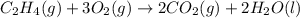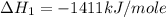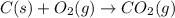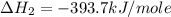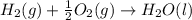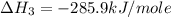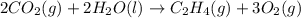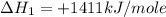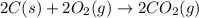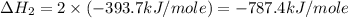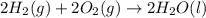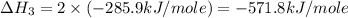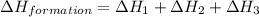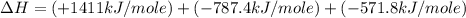Chemistry, 31.08.2019 04:10 isabella4141
How to draw hess' cycle for this question ?
carbon, hydrogen and ethane each burn exothermically in an excess of air. c(s) + o2(g) → co2(g) ahⓡ =-393.7 kj mol: 1. h2(g) + % o2(g) → h2o(1) ah® --285.9 kj mol. c2h4(g) + 302(g) → 2co2(g) + 2h2o(1) ah® --1411.0 kj mol? . use the data to calculate the standard enthalpy change of formation, ah® in kj mol? , of ethene at 298 k 2c(s) + 2h2(g) → c2h4(8)

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 20:30, allofthosefruit
Draw a line graph showing the relationship between temperature in kelvin as a function of kinetic energy.
Answers: 3


Chemistry, 23.06.2019 09:20, annapittbull12
1) a. water molecule breaks up into hydrogen and oxygen on passing electricity. does this involve breaking intermolecular or intramolecular forces of attraction. explain b. on boiling water changes to water vapor. does this involve breaking intermolecular or intramolecular forces of attraction. explain methanol evaporates faster than water. contrast the intermolecular forces and the vapor pressures of methanol and water?
Answers: 2
You know the right answer?
How to draw hess' cycle for this question ?
carbon, hydrogen and ethane each burn exothermica...
carbon, hydrogen and ethane each burn exothermica...
Questions in other subjects:

Physics, 07.01.2021 21:00





Mathematics, 07.01.2021 21:00


Biology, 07.01.2021 21:00

Health, 07.01.2021 21:00

 will be,
will be,