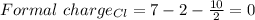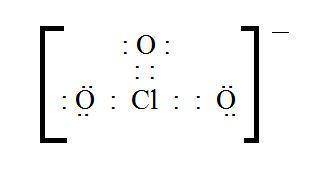Chemistry, 30.08.2019 16:20 evelyngarcia99
C. what is the formal charge of the chlorine atom in the chlorate ion (cio3)? +2, +2, 0, -1, -2

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:20, rex1578
Part b: study of equilibrium on solubility: mg(oh)2(s) ⇌ mg2+(aq) + 2 oh–(aq) cloudy clear (pink) 7. a. b. 8. a. b. 9. 10. 11. 12. when adding concentrated hydrochloric acid, how did the appearance of the equilibrium mixture change? the change in appearance indicated a shift in the point of equilibrium. in which direction did the equilibrium shift? (l) left (r) right explain your answer to question 7a. you should indicate which ion was added to or removed from the equilibrium mixture. when adding edta, how did the appearance of the equilibrium mixture change? the change in appearance indicated a shift in the point of equilibrium. in which direction did the equilibrium shift? (l) left (r) right explain your answer to question 8a. you should indicate which ion was added to or removed from the equilibrium mixture. upon heating in which direction is the equilibrium shifting? upon cooling in which direction is the equilibrium shifting? is the forward reaction a. endothermic explain your answers to questions 9, 10, and 11. (l) left (r) right (l) left (r) right b. exothermic
Answers: 1


Chemistry, 22.06.2019 22:30, xlebrny7831
Amedication is given at a dosage of 3.000 mg of medication per kg of body weight. if 0.1500 g of medication is given, then what was the patient's weight in pounds (lbs)? there are 453.59g in 1 lb.
Answers: 2
You know the right answer?
C. what is the formal charge of the chlorine atom in the chlorate ion (cio3)? +2, +2, 0, -1, -2...
Questions in other subjects:

Mathematics, 08.09.2021 01:00


Mathematics, 08.09.2021 01:00


Mathematics, 08.09.2021 01:00


Arts, 08.09.2021 01:00

Engineering, 08.09.2021 01:00

Mathematics, 08.09.2021 01:00

Mathematics, 08.09.2021 01:00

 ) :
) :