
Chemistry, 29.08.2019 20:20 jessnolonger
Carbon, hydrogen and ethane each burn exothermically in an excess of air. ahⓡ =-393.7 kj mol. c(s) + o2(g) → co2(g) h2(g) + % o2(g) → h20(1) czha(g) + 302() → 2co2(g) + 2h2o(1) ah®=-285.9 kj mol ah =-1411.0 kj moll. use the data to calculate the standard enthalpy change of formation, ah in kj mol'', of ethene at 298 k 2c(s) + 2h2(g) → c2h4(g)

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 13:00, kevinhill185
Pauling and lewis questioned the extreme definitions of bonds. they wondered if bonds might be described somewhere in between the two extremes (covalent and ionic). on the basis of experimental data, pauling confirmed that bonds could be ionic, covalent, and for those, in between, exhibit a degree of ionic character. he theorized that the major factor was how strongly the atoms in the bond attracted the electrons. pauling called this factor - the tendency of an atom to attract electrons in a bond.
Answers: 2

Chemistry, 22.06.2019 13:10, Jana1517
The last few miles of the marathon are the most difficult for heather, her hair plastered to her head, sweat clinging to her arms, and her legs already feeling as if they had nothing left, just dead weight. after grabbing a cup of ice water, she feels the ice cubes smash against her nose as she gulps some cool refreshment and keeps on running. in these last few miles, the breeze kicks up and she finally feels some coolness against her skin. drips of sweat, once clinging to her forehead, now spill down, and heather feels more pain as the sweat flows into her eyes. which of the following is the most likely reason why the ice struck heather’s nose when she took a drink? a) water can function as a solvent. b) water can store large amounts of heat. c) water can moderate temperatures through evaporative cooling. d) the density of water decreases when it freezes. e) water has a cohesive nature. sweat remained on heather’s forehead and arms because of the a) high salt content of sweat b) cohesive nature of water c) ability of water to moderate heat d) high evaporative cooling effect of water e) ability of water to act as a solvent
Answers: 1

Chemistry, 22.06.2019 18:00, meowmeowcow
Find the mass, in grams, of 5.00*10^23 molecules of f2
Answers: 3

Chemistry, 22.06.2019 22:00, Porciabeauty6788
The diagrams to the right show the distribution and arrangement of gas particles in two different containers. according to kinetic-molecular theory, which of the following statements is true? check all that apply. if the temperatures of both containers are equal, container a has greater pressure than container b. if the volume of container a decreased, its pressure would decrease. if the pressure in both containers is equal, container a has a lower temperature than container b.
Answers: 2
You know the right answer?
Carbon, hydrogen and ethane each burn exothermically in an excess of air. ahⓡ =-393.7 kj mol. c(s) +...
Questions in other subjects:


Chemistry, 04.01.2021 14:50

English, 04.01.2021 14:50

Mathematics, 04.01.2021 14:50

Physics, 04.01.2021 14:50

Mathematics, 04.01.2021 14:50

Biology, 04.01.2021 14:50

Chemistry, 04.01.2021 14:50

French, 04.01.2021 14:50

Mathematics, 04.01.2021 14:50

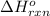 for the reaction is 51.8 kJ.
for the reaction is 51.8 kJ.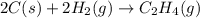
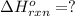
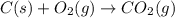
 ( × 2)
( × 2)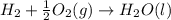
 ( × 2)
( × 2)

![\Delta H^o_{rxn}=[2\times \Delta H_1]+[2\times \Delta H_2]+[1\times (-\Delta H_3)]](/tpl/images/0209/7515/e45ac.png)
![\Delta H^o_{rxn}=[(2\times (-393.7))+(2\times (-285.9))+(1\times -(-1411))]=51.8kJ](/tpl/images/0209/7515/5c6c6.png)


