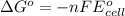Chemistry, 22.08.2019 21:30 trosclairozlynn02
Using the following standard reduction potentials, fe3+(aq) + e- → fe2+(aq) e° = +0.77 v ni2+(aq) + 2 e- → ni(s) e° = -0.23 v calculate the standard cell potential for the galvanic cell reaction given below, and determine whether or not this reaction is spontaneous under standard conditions. ni2+(aq) + 2 fe2+(aq) → 2 fe3+(aq) + ni(s)

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:30, jescanarias22
What’s the scientific notation for the number 6,840,000,000
Answers: 1

Chemistry, 22.06.2019 08:00, danielhall
Identify a strong intermolecular force of attraction between an alcohol
Answers: 1
You know the right answer?
Using the following standard reduction potentials, fe3+(aq) + e- → fe2+(aq) e° = +0.77 v ni2+(aq) +...
Questions in other subjects:



Biology, 30.11.2019 20:31



English, 30.11.2019 20:31

Mathematics, 30.11.2019 20:31

History, 30.11.2019 20:31


Chemistry, 30.11.2019 20:31



 of the reaction, we use the equation:
of the reaction, we use the equation: