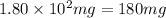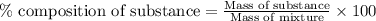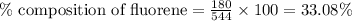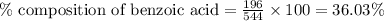A544 mg of a mixture of fluorene and benzoic acid was weighed out and subjected to an extraction and recrystallization. after this purification was completed the product crystals were dried and analyzed. the purification procedure produced 1.80 × 102 mg of fluorene and 196 mg of benzoic acid. calculate the percent composition of this mixture.

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 22:30, angelagonzalesownus1
Which statement best summarizes the importance of ernest rutherford’s gold foil experiment? it proved that all of john dalton’s postulates were true. it verified j. j. thomson’s work on the atomic structure. it showed that an electron circles a nucleus in a fixed-energy orbit. it showed that a nucleus occupies a small part of the whole atom.
Answers: 1


Chemistry, 23.06.2019 07:00, jboii11
An unknown substance is a white solid at room temperature and has a melting point of 78 °c. which of the following substances is most likely to be the identity of the unknown sample? a. naphthalene, a molecular solid with the formula c10h8 b. silica, a network solid held together by covalent bonds with the formula sio2 c. calcium chloride, an ionic compound with the formula cacl2 d. water, an molecular compound with the formula h2o
Answers: 2
You know the right answer?
A544 mg of a mixture of fluorene and benzoic acid was weighed out and subjected to an extraction and...
Questions in other subjects:

Mathematics, 29.01.2020 07:40

Social Studies, 29.01.2020 07:40

Mathematics, 29.01.2020 07:40






History, 29.01.2020 07:40

Mathematics, 29.01.2020 07:40