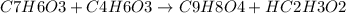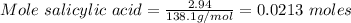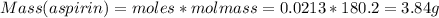Chemistry, 21.08.2019 22:30 hooplikeapro
What is the theoretical yield of aspirin (180.2 g/mol) in the reaction of 2.94 grams of salicylic acid (138.1 g/mol) with excess acetic anhydride (102.1 g/mol)? show your work. include units and round your final answer appropriately.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 19:00, georgesarkes12
Mercury metal is poured into a graduated cylinder that holds exactly 22.5 ml the mercury used to fill the cylinder mass in 306.0 g from this information calculate the density of mercury
Answers: 2

Chemistry, 22.06.2019 20:00, Isaiahtate053
The volume of a single vanadium atom is 9.29×10-24 cm3. what is the volume of a vanadium atom in microliters?
Answers: 3


Chemistry, 23.06.2019 00:20, jessicamcummins
What type of context clue you understand the meaning of quandary?
Answers: 3
You know the right answer?
What is the theoretical yield of aspirin (180.2 g/mol) in the reaction of 2.94 grams of salicylic ac...
Questions in other subjects:


Biology, 21.11.2019 22:31



Mathematics, 21.11.2019 22:31



Mathematics, 21.11.2019 22:31

Biology, 21.11.2019 22:31