
Asolution is 0.010 m in each of pb(no3)2, mn(no3)2, and zn(no3)2. solid naoh is added until the ph of the solution is 8.50. which of the following statements is true? salt ksp pb(oh)2 1.4 × 10–20 mn(oh)2 2.0 × 10–13 zn(oh)2 2.1 × 10–16 a. all three hydroxides will precipitate. b. no precipitate will form. c. only pb(oh)2 will precipitate. d. only mn(oh)2 will precipitate. e. only zn(oh)2 and pb(oh)2 will precipitate.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 15:00, emmalie52
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. p k a1 p k a2 1.30 6.70 calculate the ph for each of the points in the titration of 50.0 ml of 1.5 m h3po3(aq) 1.5 m h 3 po 3 ( aq ) with 1.5 m koh(aq). 1.5 m koh ( aq ) .
Answers: 1

Chemistry, 22.06.2019 16:00, hjgjlgkjg
Inside a flashbulb, oxygen surrounds a thin coil of magnesium. when the flashbulb is set off, a chemical reaction takes place in which magnesium combines with oxygen to form magnesium oxide. which of the chemical equations matches the reaction above? a. mg + o2 mgo2 + energy b. 2mg + o mg2o + energy c. 2mg + o2 2mgo + energy d. mg + o mgo + energy
Answers: 1

You know the right answer?
Asolution is 0.010 m in each of pb(no3)2, mn(no3)2, and zn(no3)2. solid naoh is added until the ph o...
Questions in other subjects:










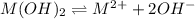
![K_{sp}=[M^{2+}][OH^{-}]^{2}](/tpl/images/0183/5838/c78d3.png)
![[M^{2+}]](/tpl/images/0183/5838/437cc.png) and
and ![[OH^{-}]^{2}](/tpl/images/0183/5838/53c7a.png) exceeds
exceeds  values of given hydroxides then hydroxides of given metal ions will precipitate.
values of given hydroxides then hydroxides of given metal ions will precipitate.![-log[OH^{-}]](/tpl/images/0183/5838/04732.png) = 5.50
= 5.50![[OH^{-}]](/tpl/images/0183/5838/e46dd.png) =
= 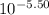

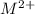 ) are 0.010 M
) are 0.010 M![[M^{2+}][OH^{-}]^{2}=0.010\times (3.16\times 10^{-6})^{2}=3.16\times 10^{-8}](/tpl/images/0183/5838/d1788.png)



