
Chemistry, 20.08.2019 23:30 ricardorendon100
Consider the following reaction where kc = 154 at 298 k.2no(g) + br2(g) 2nobr(g)a reaction mixture was found to contain 4.64×10-2 moles of no(g), 4.56×10-2 moles of br2(g) and 0.102 moles of nobr(g), in a 1.00 liter container. is the reaction at equilibrium? if not, what direction must it run in order to reach equilibrium? the reaction quotient, qc, equals .the reactiona. must run in the forward direction to reach equilibrium. b. must run in the reverse direction to reach equilibrium. c. is at equilibrium.

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 21.06.2019 23:30, huangjianhe135
Start an single atom tab. observe the decay of polonium-211. after each decay, press the reset nucleus button to watch the process again. write a description of alpha decay for po-211
Answers: 2


Chemistry, 22.06.2019 08:00, ggdvj9gggsc
Asap! will give brainiest when a heat wave strikes a region causing more people to run air-conditioning units, electrical demand increases. what needs to be done to meet this increased demand? raising the control rodslowering the control rodsremoving the control rods
Answers: 1
You know the right answer?
Consider the following reaction where kc = 154 at 298 k.2no(g) + br2(g) 2nobr(g)a reaction mixture w...
Questions in other subjects:


Mathematics, 25.06.2020 02:01


Computers and Technology, 25.06.2020 02:01





Mathematics, 25.06.2020 02:01

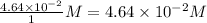
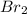 =
= 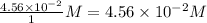
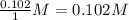
 , for this reaction =
, for this reaction = ![\frac{[NOBr]^{2}}{[NO]^{2}[Br_{2}]}](/tpl/images/0183/0904/2c141.png)
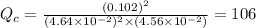
 therefore reaction must run in forward direction to increase
therefore reaction must run in forward direction to increase  .
.

