
Chemistry, 20.08.2019 22:20 davionb556
Consider the following reaction where kc = 1.80×10-2 at 698 k: 2 hi (g) h2 (g) + i2 (g)a reaction mixture was found to contain 0.304 moles of hi (g), 5.07×10-2 moles of h2 (g), and 4.57×10-2 moles of i2 (g), in a 1.00 liter container. indicate true (t) or false (f) for each of the following: 1. in order to reach equilibrium hi(g) must be produced .2. in order to reach equilibrium kc must increase .3. in order to reach equilibrium h2 must be consumed .4. qc is less than kc.5. the reaction is at equilibrium. no further reaction will occur.

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 21.06.2019 21:30, awdadaddda
How air particles exert a pressure on the inside of the balloon
Answers: 1


You know the right answer?
Consider the following reaction where kc = 1.80×10-2 at 698 k: 2 hi (g) h2 (g) + i2 (g)a reaction mi...
Questions in other subjects:

Arts, 27.01.2021 22:30

Mathematics, 27.01.2021 22:30


Arts, 27.01.2021 22:30


Mathematics, 27.01.2021 22:30




Social Studies, 27.01.2021 22:30

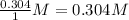
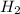 =
= 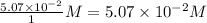
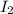 =
= 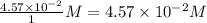
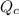 , for this reaction =
, for this reaction = ![\frac{[H_{2}][I_{2}]}{[HI]^{2}}](/tpl/images/0182/8823/7ae06.png)
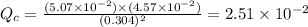
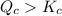 therefore reaction must run in reverse direction to reduce
therefore reaction must run in reverse direction to reduce 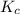 . That means HI(g) must be produced and
. That means HI(g) must be produced and 

