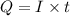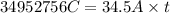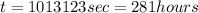Purification of copper can be achieved by electrorefining copper from an impure copper anode onto a pure copper cathode in an electrolytic cell. how many hours will it take to plate 11.5 kg of copper onto the cathode if the current passed through the cell is held constant at 34.5 a ? assume the copper in the electrolytic solution is present as cu2+ .

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:30, salvadorperez26
Suppose that during that icy hot lab 65,000 j of energy were transferred to 450 g of water at 20°c what would have have been the final temperature of the water
Answers: 2

Chemistry, 22.06.2019 08:30, microwave13016
Agroup of students is studying convection current. they fill two identical balloons with the same amount of helium. one balloon is placed in a freezer and the other is in an area with warm air. after 10 minutes, the balloon are released from a height of 1 meter. which of the following to the students most likely observe? a) the warm balloon expands and rises. the cold balloon shrinks and sinks b) the balloon both rise. the cold balloon is larger than the warm balloon c) the cold balloon expands and rises. the warm balloon shrinks and sinks d) the balloon rise at the same rate. both balloons are the same size
Answers: 1

You know the right answer?
Purification of copper can be achieved by electrorefining copper from an impure copper anode onto a...
Questions in other subjects:

Mathematics, 12.02.2021 21:20






History, 12.02.2021 21:20

Physics, 12.02.2021 21:20

Mathematics, 12.02.2021 21:20

Mathematics, 12.02.2021 21:20

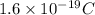
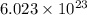 electrons
electrons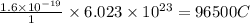
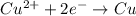
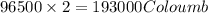 of electricity deposits 1 mole or 63.5 g of copper
of electricity deposits 1 mole or 63.5 g of copper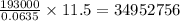 Coloumb
Coloumb