
Chemistry, 17.08.2019 19:10 brittanyfox411
A25.00-ml sample of propionic acid, hc3h5o2, of unknown concentration was titrated with 0.141 m koh. the equivalence point was reached when 43.76 ml of base had been added. what is the hydroxide-ion concentration at the equivalence point? ka for propionic acid is 1.3 × 10–5 at 25°c. a. 1.5 × 10-9 m b. 1.1 × 10-3 m c. 1.1 × 10-5 m d. 8.3 × 10-6 m e. 1.0 × 10-7 m

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 16:30, joshua1255
Find the number of moles of argon in 364g of argon.
Answers: 2

Chemistry, 23.06.2019 11:00, landon6663
Which of the following reactions represents an exothermic reaction? nh3(g) + 12.0 kcal ½n2(g) + 3/2 h2(g) ch4 + 2o2 co2 + 2h2o + 212,800 cal c + 2s cs2, h = 27,550 cal c(graphite) c(diamond), h = 0.45 kcal 2h2o 2h2 + o2, h = +58 kcal
Answers: 1


Chemistry, 23.06.2019 13:30, michelle8978
Why hydrochloric acid neutralized first when you titrate a mixture of hcl& ch3cooh against standard sodium hydroxide
Answers: 1
You know the right answer?
A25.00-ml sample of propionic acid, hc3h5o2, of unknown concentration was titrated with 0.141 m koh....
Questions in other subjects:



English, 26.09.2019 01:30

Mathematics, 26.09.2019 01:30


Biology, 26.09.2019 01:30

Health, 26.09.2019 01:30

Mathematics, 26.09.2019 01:30


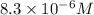
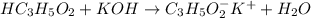
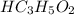 reacts with 1 mol of KOH to produce 1 mol of
reacts with 1 mol of KOH to produce 1 mol of 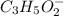
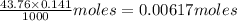
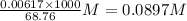
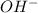 produced at equivalence point is due to hydrolysis of
produced at equivalence point is due to hydrolysis of 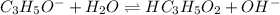
![\frac{[HC_{3}H_{5}O_{2}][OH^{-}]}{[C_{3}H_{5}O_{2}^{-}]}=K_{b}(C_{3}H_{5}O_{2}^{-})=\frac{10^{-14}}{K_{a}(HC_{3}H_{5}O_{2})}](/tpl/images/0175/3912/dc120.png)
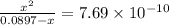
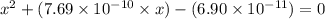
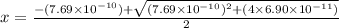 M =
M = 

