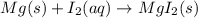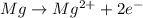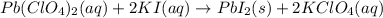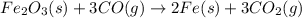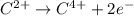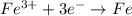Chemistry, 16.08.2019 08:10 avastanleyy
Which of the following chemical equations show(s) oxidation-reduction reactions? 1. mg(s) + i2(aq) --> mgi2(s)2. pb(clo4)2(aq) + 2 ki(aq) --> pbi2(s) + 2 kclo4(aq)3. fe2o3(s) + 3 co(g) --> 2 fe(s) + 3 co2(g)1 only2 only1 and 21 and 32 and 3

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 05:30, maddyjones4172
Which of the following two events occur to create a sea breeze? select all that apply. warm air rises on the ocean and moves toward the land to cool warm air rises on land and moves toward the ocean to cool cool air moves from the ocean to be warmed by the land cool air moves from the land to be warmed by the ocean
Answers: 3

Chemistry, 22.06.2019 14:30, joejoefofana
Consider the reduction reactions and their equilibrium constants. cu+(aq)+e−↽−−⇀cu(s)pb2+(aq)+2e−↽−−⇀ pb(s)fe3+(aq)+3e−↽−−⇀fe(=6.2×108=4. 0×10−5=9.3×10−3 cu + ( aq ) + e − ↽ − − ⇀ cu ( s ) k =6.2× 10 8 pb 2 + ( aq ) +2 e − ↽ − − ⇀ pb ( s ) k =4.0× 10 − 5 fe 3 + ( aq ) +3 e − ↽ − − ⇀ fe ( s ) k =9.3× 10 − 3 arrange these ions from strongest to weakest oxidizing agent.
Answers: 3

Chemistry, 22.06.2019 17:00, smelcher3900
According to the kinetic-molecular theory, what happens to a liquid when it is transferred from one container to another? the volume and the shape stay the same. the volume increases to fill the new container, but the shape stays the same. the volume stays the same, but the shape changes to fit the new container. the volume and the shape change to fill the new container.
Answers: 2
You know the right answer?
Which of the following chemical equations show(s) oxidation-reduction reactions? 1. mg(s) + i2(aq) -...
Questions in other subjects:

Mathematics, 08.07.2019 21:00


History, 08.07.2019 21:00

History, 08.07.2019 21:00

Mathematics, 08.07.2019 21:00

Mathematics, 08.07.2019 21:00

Mathematics, 08.07.2019 21:00

Chemistry, 08.07.2019 21:00

Biology, 08.07.2019 21:00

Social Studies, 08.07.2019 21:00