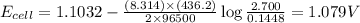Consider the reaction corresponding to a voltaic cell and its standard cell potential. z n ( s ) + c u 2 + ( a q ) ⟶ c u ( s ) + z n 2 + ( a q ) zn(s)+cux2+(aq)⟶cu(s)+znx2+(aq) e o cell = 1.1032 v ecello=1.1032 v what is the cell potential for a cell with a 2.700 m solution of z n 2 + ( a q ) znx2+(aq) and 0.1448 m solution of c u 2 + ( a q ) cux2+(aq) at 436.2 k? c

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:30, earcake2470
How many grams of co(g) are there in 74.5 ml of the gas at 0.933 atm and 30o c?
Answers: 1

Chemistry, 22.06.2019 12:00, angtrevv
In a laboratory, 1.55mg of an organic compound containing carbon, hydrogen, and oxygen is burned for analysis. this combustion resulted in the formation of 1.45mg of carbon dioxide and .89 mg of water. what is the empirical formula for this compound?
Answers: 1

Chemistry, 22.06.2019 12:20, jessicasbss6840
Adeuteron, 21h, is the nucleus of a hydrogen isotope and consists of one proton and one neutron. the plasma of deuterons in a nuclear fusion reactor must be heated to about 3.02×108 k . what is the rms speed of the deuterons? express your answer using two significant figures.
Answers: 1

Chemistry, 22.06.2019 16:30, Eddie997
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2
You know the right answer?
Consider the reaction corresponding to a voltaic cell and its standard cell potential. z n ( s ) + c...
Questions in other subjects:

English, 05.12.2021 19:10


English, 05.12.2021 19:10

English, 05.12.2021 19:10


Chemistry, 05.12.2021 19:10

History, 05.12.2021 19:10

Mathematics, 05.12.2021 19:10

Health, 05.12.2021 19:10


![E_{cell}=E^o_{cell}-\frac{RT}{nF}\log \frac{[Zn^{2+}]^2}{[Cu^{2+}]}](/tpl/images/0174/9065/fa75b.png)
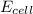 = cell potential of the cell = ?
= cell potential of the cell = ?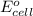 = standard electrode potential = 1.1032 V
= standard electrode potential = 1.1032 V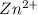 = 2.700 M
= 2.700 M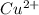 = 0.1448 M
= 0.1448 M