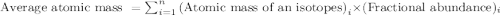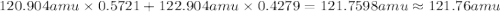Chemistry, 13.08.2019 06:10 QuestionsAnsweredNow
The two naturally occurring isotopes of antimony, 121sb (57.21 percent) and 123sb (42.79 percent), have masses of 120.904 and 122.904 amu, respectively. what is the average atomic mass of sb? (a) 121.90 amu (b) 122.05 amu (c) 121.76 amu (d) 121.34 amu (e) 122.18 amu

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:30, krharris
Melting and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 2

Chemistry, 22.06.2019 10:30, angemango3423
What is the empirical formula of c6h18o3? ch3o c2h5o c2h6o c2h5o5
Answers: 1

Chemistry, 22.06.2019 15:10, strodersage
Which statement describes the phase change that occurs when dry ice is placed in an open container at room temperature?
Answers: 1

Chemistry, 22.06.2019 21:00, nsutton9985
Two nails have identical sizes and shapes. in one nail, 20 percent of the domains are lined up. in the other nail, 80 percent of the domains are lined up. which has stronger magnetic force? first answer gets brainliest!
Answers: 1
You know the right answer?
The two naturally occurring isotopes of antimony, 121sb (57.21 percent) and 123sb (42.79 percent), h...
Questions in other subjects:


Computers and Technology, 28.08.2019 20:30

Computers and Technology, 28.08.2019 20:30


Computers and Technology, 28.08.2019 20:30