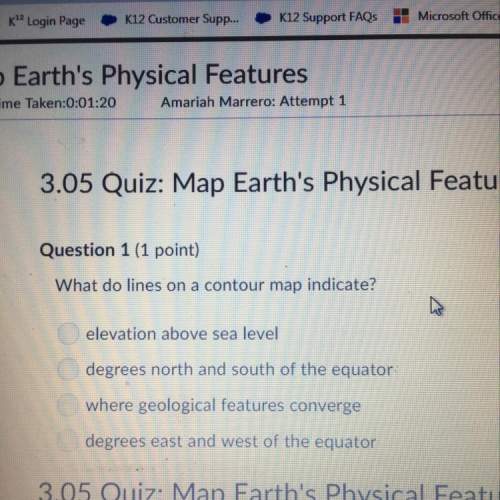
:(10 points) a brine solution of salt flows at a constant rate of 4 l/min into a large tank that initially held 100 l of pure water. the solution inside the tank is kept well stirred and flows out of the tank at a rate of 3 l/min. if the concentration of the salt in the brine entering the tank is 0.2 kg/l, determine the mass of salt in the tank after t min.

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 07:30, gwenparks
Calculate the ratio of h+ ions to oh– ions at a ph = 7. find the concentration of h+ ions to oh– ions listed in table b of your student guide. then divide the h+ concentration by the oh– concentration. record this calculated ratio in table a of your student guide. compare your approximated and calculated ratios of h+ ions to oh– ions at a ph = 7. are they the same? why or why not? record your comparison in table a. what is the concentration of h+ ions at a ph = 7? mol/l what is the concentration of oh– ions at a ph = 7? mol/l what is the ratio of h+ ions to oh– ions at a ph = 7? : 1
Answers: 1

Chemistry, 22.06.2019 08:30, ebigham5117
If i initially have a gas at a pressure of 12 atm, a volume of 23 liters, and a temperature of 200 k, and then i raise the pressure to 14 atm and increase the temperature to 300 k, what is the new volume of the gas?
Answers: 2

Chemistry, 22.06.2019 11:50, tajanaewilliams77
If oil spills continue, all of the following should be expected except (2 points) death of aquatic life. polluted groundwater. decreased soil productivity. increased global temperatures.
Answers: 3
You know the right answer?
:(10 points) a brine solution of salt flows at a constant rate of 4 l/min into a large tank that ini...
Questions in other subjects:

Computers and Technology, 22.02.2021 06:10

Mathematics, 22.02.2021 06:10

Mathematics, 22.02.2021 06:10

English, 22.02.2021 06:10

English, 22.02.2021 06:10



History, 22.02.2021 06:10


English, 22.02.2021 06:10