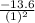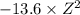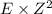Chemistry, 13.08.2019 03:20 skyhighozzie
Energy levels: an ionized atom having z protons has had all but one of its electrons removed. if e is the total energy of the ground state electron in atomic hydrogen, then what is the total energy of the remaining electron in the ionized atom?

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 12:30, nekathadon
The bond energy for the van der waals bond between two helium atoms is 7.9×10−4ev. assuming that the average kinetic energy of a helium atom is (3/2)kbt, at what temperature is the average kinetic energy equal to the bond energy between two helium atoms
Answers: 1

Chemistry, 22.06.2019 21:30, shiannethorn
How many liters of 3.0 m naoh solution will react with 0.60 liters of 4.0 m h2so4? h2so4 + naoh → na2so4 + h2o 1.2 l 1.6 l 2.4 l 2.8 l
Answers: 3

Chemistry, 23.06.2019 00:20, destromero
Which diagram represents the phase tha occurs after a solid melts?
Answers: 1

Chemistry, 23.06.2019 00:20, jessicamcummins
What type of context clue you understand the meaning of quandary?
Answers: 3
You know the right answer?
Energy levels: an ionized atom having z protons has had all but one of its electrons removed. if e...
Questions in other subjects:


Mathematics, 12.08.2020 17:01



Mathematics, 12.08.2020 17:01





Mathematics, 12.08.2020 17:01

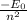
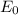 = 13.6
= 13.6