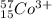Write the symbol for each of the following ions:
(a) the ion with a 1+ charge, atomic number 55, and mass number 133
(b) the ion with 54 electrons, 53 protons, and 74 neutrons
(c) the ion with atomic number 15, mass number 31, and a 3 - charge
(d) the ion with 24 electrons, 30 neutrons, and a 3+ charge

Answers: 2


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 15:10, strodersage
Which statement describes the phase change that occurs when dry ice is placed in an open container at room temperature?
Answers: 1
You know the right answer?
Write the symbol for each of the following ions:
(a) the ion with a 1+ charge, atomic number...
(a) the ion with a 1+ charge, atomic number...
Questions in other subjects:







Mathematics, 04.11.2019 23:31


History, 04.11.2019 23:31

Mathematics, 04.11.2019 23:31

 .
.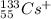 .
.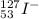 .
.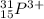 .
.