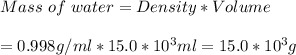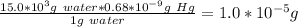Chemistry, 12.08.2019 18:30 jujulakaeuaws
The mercury content of a stream was belived to be above the minimum considered safe part per billion (ppb) by weight. an analysis indicated that the concentration was 0.68 parts per billion. what quantity of mercury in grams was persent in 15.0 l of the water, the density of which is 0.998 g/ml? (1 ppb hg = 1nghg/1g water)

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 12:00, BreBreDoeCCx
Ageochemist examines a piece of metal that he found in the soil. he performs tests to identify the metal from its density, electrical conductivity, and melting point. which statement best describes his investigation? a. he is determining physical properties that are sufficient to identify the metal. b. he is determining chemical properties that are sufficient to identify the metal. c. he is determining physical properties that are insufficient to identify the metal. d. he is determining chemical properties that are insufficient to identify the metal.
Answers: 3

Chemistry, 23.06.2019 01:00, jazzy200232
Which process results in the release of energy stored in the products of photosynthesis? a. polymer synthesis b. depolymerization c. digestion d. cellular respiration
Answers: 1


Chemistry, 23.06.2019 02:50, igraha17
Dumbledore decides to gives a surprise demonstration. he starts with a hydrate of na2co3 which has a mass of 4.31 g before heating. after he heats it he finds the mass of the anhydrous compound is found to be 3.22 g. he asks everyone in class to determine the integer x in the hydrate: na2co3·xh2o; you should do this also. round your answer to the nearest integ
Answers: 2
You know the right answer?
The mercury content of a stream was belived to be above the minimum considered safe part per billio...
Questions in other subjects:

Social Studies, 29.07.2019 18:00

Biology, 29.07.2019 18:00

History, 29.07.2019 18:00

Biology, 29.07.2019 18:00

Mathematics, 29.07.2019 18:00





Mathematics, 29.07.2019 18:10