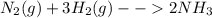Chemistry, 12.08.2019 18:20 baylabrown
Nitrogen (n2) and hydrogen (h2) react to form ammonia (nh3). consider a mixture of six nitrogen molecules and six hydrogen molecules in a closed container. assuming the reaction goes to completion, what will the final product mixture be

Answers: 1


Other questions on the subject: Chemistry



Chemistry, 23.06.2019 00:00, maronetham6253
What is the empirical formula of a compound that is 50.7% antimony and 49.3% selenium ?
Answers: 2
You know the right answer?
Nitrogen (n2) and hydrogen (h2) react to form ammonia (nh3). consider a mixture of six nitrogen mole...
Questions in other subjects:


Biology, 31.08.2020 20:01





Mathematics, 31.08.2020 20:01


Mathematics, 31.08.2020 20:01

Spanish, 31.08.2020 20:01