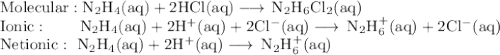Chemistry, 09.08.2019 01:20 zenaidazurita1p6bs1d
Write the molecular, ionic, and net ionic equations for the reaction of a: hcl with koh, b: hcho2 with lioh and c: n2h4 with hcl. include states in your answer.

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 21.06.2019 23:30, huangjianhe135
Start an single atom tab. observe the decay of polonium-211. after each decay, press the reset nucleus button to watch the process again. write a description of alpha decay for po-211
Answers: 2

Chemistry, 22.06.2019 17:00, brownvester44
Astable electron arrangement for an atom is one that does not easily change. how is this arrangement arrived at? a. valence electrons are transferred or shared to create a full outer shell of electrons. b. valence electrons are discarded into space to create a full outer shell of electrons. c. protons (positive charge) pair with valence electrons (negative charge) to create a strong bond. d. outer shells with valence electrons are transferred or shared.
Answers: 2

Chemistry, 22.06.2019 19:20, johnkings140
For a research project, a student decided to test the effect of the lead(ii) ion (pb2+) on the ability of salmon eggs to hatch. this ion was obtainable from the water‐soluble salt, lead(ii) nitrate, which the student decided to make by the following reaction. pbo(s) + 2 hno3(aq) → pb(no3)2(aq) + h2o losses of product for various reasons were expected, and a yield of 86.0% was expected. in order to have 5.00 g of product at this yield, how many grams of pbo should be reacted? (assume that sufficient nitric acid, hno3, would be used.)
Answers: 1
You know the right answer?
Write the molecular, ionic, and net ionic equations for the reaction of a: hcl with koh, b: hcho2...
Questions in other subjects:

Mathematics, 18.09.2020 20:01

Mathematics, 18.09.2020 20:01

Mathematics, 18.09.2020 20:01

Mathematics, 18.09.2020 20:01

Biology, 18.09.2020 20:01

Mathematics, 18.09.2020 20:01

Geography, 18.09.2020 20:01

Mathematics, 18.09.2020 20:01

History, 18.09.2020 20:01

Mathematics, 18.09.2020 20:01