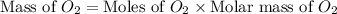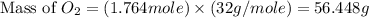Chemistry, 06.08.2019 05:30 lpslover26237
Glucose, c6h12o6, is used as an energy source by the human body. the overall reaction in the body is described by the equation c6h12o6(aq)+6o2(g)⟶6co2(g)+6h2o(l)
calculate the number of grams of oxygen required to convert 53.0 g of glucose to co2 and h2o.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 12:10, yootmytoot
Achemistry student needs to standardize a fresh solution of sodium hydroxide. he carefully weighs out of oxalic acid , a diprotic acid that can be purchased inexpensively in high purity, and dissolves it in of distilled water. the student then titrates the oxalic acid solution with his sodium hydroxide solution. when the titration reaches the equivalence point, the student finds he has used of sodium hydroxide solution. calculate the molarity of the student's sodium hydroxide solution. be sure your answer has the correct number of significant digits.
Answers: 1


Chemistry, 22.06.2019 19:30, 2020sanchezyiczela
Draw the lewis structure for the trisulfur s3 molecule. be sure to include all resonance structures that satisfy the octet rule.
Answers: 3

Chemistry, 23.06.2019 08:40, Riplilpeep
Which statement is true according to the kinetic theory? a. molecules of different gases with the same mass and temperature always have the same average density. b. molecules of different gases with the same mass and temperature always have the same average volume. c. molecules of different gases with the same mass and temperature always have the same pressure. d. molecules of different gases with the same mass and temperature always have the same molecular mass. e. molecules of different gases with the same mass and temperature always have the same average kinetic energy.
Answers: 1
You know the right answer?
Glucose, c6h12o6, is used as an energy source by the human body. the overall reaction in the body is...
Questions in other subjects:


Mathematics, 10.03.2020 22:58


Mathematics, 10.03.2020 22:58




Computers and Technology, 10.03.2020 22:58

Chemistry, 10.03.2020 22:58

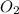 = 32 g/mole
= 32 g/mole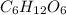 .
.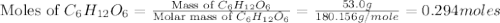
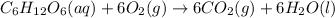
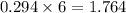 mole of
mole of