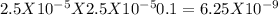Chemistry, 06.08.2019 03:20 orianabrown15
The ph of a 0.1 m mcl (m is an unknown cation) was found to be 4.7. write the net ionic equation for the hydrolysis of m and its corresponding equilibrium expression kb. calculate the value of kb

Answers: 2


Other questions on the subject: Chemistry




You know the right answer?
The ph of a 0.1 m mcl (m is an unknown cation) was found to be 4.7. write the net ionic equation for...
Questions in other subjects:

English, 20.05.2021 04:10

Mathematics, 20.05.2021 04:10



Law, 20.05.2021 04:10




Biology, 20.05.2021 04:10

Mathematics, 20.05.2021 04:10

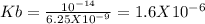

![\frac{[H^{+}][MOH]}{[M^{+}]}](/tpl/images/0172/0660/8857d.png)