
Chemistry, 01.08.2019 01:20 jademckinziemea
For the second-order reaction below, the initial concentration of reactant a is 0.24 m. if the rate constant for the reaction is 1.5 x10–2 m–1s–1, what is the concentration of a after 265 seconds? 2a --> b + crate = k[a]20.12 m0.19 m0.95 m4.0 m5.2 m

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 15:00, asims13
Which element in the third period would you expect to have the larger atomic radius, sodium (na) or sulfur (s)? a. sodium, because it has a higher effective nuclear charge attracting electrons in fewer energy levels. b. sodium, because it has fewer protons attracting electrons in the same energy levels. c. sulfur, because it has more protons attracting electrons in more energy levels. d. sulfur, because it has a higher effective nuclear charge attracting electrons in the same energy levels.
Answers: 2

Chemistry, 22.06.2019 10:00, winstonbendariovvygn
Diffraction is when light is bent around obstructions. which of the these observation about clouds would indicate diffraction? a) after rain storms, you can sometimes see rainbows. b) clouds are white or gray and cannot be seen through. c) on a cloudy day, the temperature tends to be cooler than a sunny day. d) the edges of dark clouds appear lighter. this
Answers: 3

Chemistry, 22.06.2019 14:00, claudia122752
Will mark brainliest how many electrons can be held in the energy level n = 4?
Answers: 1

Chemistry, 22.06.2019 15:00, MilanPatel
How is the shape of the poem “peer” connected to its meaning?
Answers: 2
You know the right answer?
For the second-order reaction below, the initial concentration of reactant a is 0.24 m. if the rate...
Questions in other subjects:

Mathematics, 13.12.2020 18:50

Social Studies, 13.12.2020 18:50

Social Studies, 13.12.2020 18:50

Mathematics, 13.12.2020 18:50

Chemistry, 13.12.2020 18:50

English, 13.12.2020 18:50




![\frac{1}{[A]}=\frac{1}{[A]_{0}}+kt](/tpl/images/0156/3924/a6d73.png)
![[A]_{0}](/tpl/images/0156/3924/48818.png) is intital concentration of A and k is rate constant.
is intital concentration of A and k is rate constant.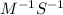 and t is 265 S
and t is 265 S![\frac{1}{[A]}=\frac{1}{0.24}+(0.015\times 265)](/tpl/images/0156/3924/91240.png)


