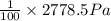The vapor pressure of pure water at 296 k is 2778.5 pa. the vapor forms an ideal gas. 1) in some oil, the equilibrium concentration of water molecules is only 1% as large as in pure water. suppose that the pure water is covered with a layer of that oil. what's the equilibrium vapor pressure of water above the oil layer?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 15:00, Homepage10
Theoretically, which metal should be the most reactive? hydrogen lithium francium fluorine
Answers: 1



Chemistry, 22.06.2019 16:50, Pookiev
Which of the following is an indication that a substance has undergone a chemical change? a. no new product has been formed. b. the color of the substance has not changed. c. the original constitute has not changed. d. the molecular structure has changed.
Answers: 1
You know the right answer?
The vapor pressure of pure water at 296 k is 2778.5 pa. the vapor forms an ideal gas. 1) in some oil...
Questions in other subjects:










 =
=