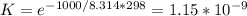Chemistry, 29.07.2019 19:20 karatekats1
Use the free energies of formation given below to calculate the equilibrium constant (k) for the following reaction at 298 k. 2 hno3(aq) + no(g) → 3 no2(g) + h2o(l) k = ? δ g0f(kj/mol) -110.9 87.6 51.3 -237.1

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 13:30, suemmimonjaras8374
The atomic number, or number, is the described as the number of in the nucleus of an chemical element.
Answers: 1

Chemistry, 22.06.2019 14:30, neidaq12345
Select the word from the list that best fits the definition the nuclear family into which a person is born or adopted.
Answers: 2

Chemistry, 22.06.2019 16:30, Kathryn014
4. a 20-kg child is tossed up into the air by her parent. the child is 2 meters off the ground traveling 5 m/s. circle one: ke / gpe / both show your work for finding the values of each type of energy the object has:
Answers: 1

Chemistry, 22.06.2019 20:00, bettybales1986
Glucose (c6h12o6) is an important biological molecule. (round the answer to nearest hundredth.) what is the percent by mass of carbon in glucose?
Answers: 2
You know the right answer?
Use the free energies of formation given below to calculate the equilibrium constant (k) for the fol...
Questions in other subjects:

Chemistry, 07.01.2020 22:31

Arts, 07.01.2020 22:31





Mathematics, 07.01.2020 22:31

Mathematics, 07.01.2020 22:31


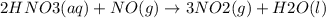
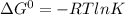

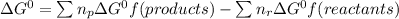
![\Delta G^{0}=[3\Delta G^{0}f(NO2)+3\Delta G^{0}f(H2O)]-[2\Delta G^{0}f(HNO3)+1\Delta G^{0}f(NO)]](/tpl/images/0147/7522/36e5a.png)
![\Delta G^{0}=[3\Delta G^{0}f(51.3)+3\Delta G^{0}f(-237.1)]-[2\Delta G^{0}f(-110.9)+1\Delta G^{0}f(87.6)]](/tpl/images/0147/7522/2150b.png)