
The equilibrium constant, kc, for the following reaction is 77.5 at 600 k. co(g) + cl2(g) cocl2(g) calculate the equilibrium concentrations of reactant and products when 0.442 moles of co and 0.442 moles of cl2 are introduced into a 1.00 l vessel at 600 k. [co] = m [cl2] = m [cocl2] = m

Answers: 1


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 18:30, tanviknawale
Which sample at stp has the same number of atoms as 18 liters of ne at stp
Answers: 1
You know the right answer?
The equilibrium constant, kc, for the following reaction is 77.5 at 600 k. co(g) + cl2(g) cocl2(g) c...
Questions in other subjects:


History, 22.05.2020 21:06

History, 22.05.2020 21:06


Health, 22.05.2020 21:06

English, 22.05.2020 21:06

English, 22.05.2020 21:06

Mathematics, 22.05.2020 21:06

Mathematics, 22.05.2020 21:06

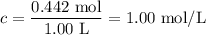
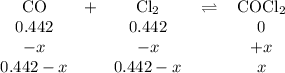
![K_{\text{c}} = \dfrac{\text{[COCl$_{2}$]}}{\text{[CO][Cl$_2$]}} = \dfrac{x}{(0.442 - x)^{2}} = 77.5\\\begin{array}{rcl}\\\dfrac{x}{(0.442 - x)^{2}} & = & 77.5\\\\x & = & 77.5(0.442 - x)^{2}\\x & = & 77.5(0.1954 - 0.884x + x^{2}\\x & = & 15.14 - 68.51x + 77.5x^{2}\\77.5x^{2} - 69.51x + 15.14 & = & 0\\x & = & \mathbf{0.373}\\\end{array}](/tpl/images/0136/6655/e9c11.png)


